Tuesday, April 30, 2019
Ractopamine

STORY AT-A-GLANCE
- The U.S. has asked China to lift a ban on U.S. pork fed the additive ractopamine, but so far China has rejected the request
- Ractopamine, a beta agonist used to increase weight gain, feed efficiency and leanness in pigs, cattle and turkeys, is banned in most countries, including China, Russia, Taiwan and the European Union
- Only 26 major meat-producing countries allow ractopamine use, while at least 160 have banned it
- The growth drug, marketed as Paylean for pigs, Optaflexx for cattle and Topmax for turkeys, is controversial not only because it’s linked to adverse effects in animals but also because of human health concerns
- The continued use of the drug in U.S. meat is causing tensions during talks aimed at ending the U.S.-China trade war
The U.S. has asked China to lift a ban on U.S. pork fed the additive ractopamine, but so far China has rejected the request. China has seen huge losses in the pork industry due to African swine fever, which may lead to the deaths of up to 200 million pigs.1
Still, even while facing protein deficits as a result, the country has been resistant to importing ractopamine-treated meat. In fact, ractopamine, a beta agonist used to increase weight gain, feed efficiency and leanness in pigs, cattle and turkeys, is banned in most other countries, including China, Russia, Taiwan and the European Union.2
Only 26 major meat-producing countries allow ractopamine use,3 while at least 160 have banned it.4The growth drug, marketed as Paylean for pigs, Optaflexx for cattle and Topmax for turkeys, is controversial not only because it's linked to adverse effects in animals but also because of human health concerns.
The continued use of the drug in U.S. meat is causing tensions during talks aimed at ending the U.S.-China trade war. According to Reuters:5
"China would likely lift a ban on U.S. poultry as part of a trade deal and may buy more pork to meet a growing supply deficit, but it is not willing to allow a prohibited growth drug used in roughly half the U.S. hog herd, two sources with knowledge of the negotiations said."
Ractopamine Mimics Stress Hormones, Leads to Health Problems in Pigs
The U.S. Food and Drug Administration (FDA) approved the use of ractopamine in feed for pigs in 1999, later adding in approval for cattle and turkeys. The approval was based largely on studies conducted by the drug's maker, Elanco, which focused not on safety for the animals or humans, but rather on economics, including what dosage to administer to raise bigger animals, faster.6
According to the Center for Food Safety (CFS), "The drug mimics stress hormones and increases the rate at which the animals convert feed to muscle."7 In so doing, however, a number of adverse effects have been reported in pigs, including high stress levels, lameness, hyperactivity, broken limbs and death.
Problems with behavior and cardiovascular, musculoskeletal, reproductive and endocrine systems have also been reported.
"Fed to an estimated 60 to 80 percent of pigs in the U.S. meat industry, ractopamine use has resulted in more reports of sickened or dead pigs than any other livestock drug on the market," CFS noted. "According to FDA's own calculations, more pigs have been adversely affected by ractopamine than by any other animal drug — more than 160,000."8
One study of ractopamine in monkeys showed the animals developed rapid heartbeat, whereas rats developed a host of birth defects ranging from cleft palate and short limbs to open eyelids and enlarged heart.9,10
Further, a study published in Translational Animal Science reported that the use of ractopamine has been associated with increased risks of nonambulatory and injured pigs, increased cattle mortality on feedlots and abnormal cases of hoof sloughing at beef packing plants.11 In short, hoof problems appear common in cattle taking the drug and related beta agonist drugs such as zilpateral (Zilmax), including the outer shell of the hoof falling off.
Pigs taking the drug are more likely to lose the ability to walk and become more difficult to handle and transport, succumbing more easily to stress as well.12 Ractopamine-treated pigs may become more aggressive and are more likely to be handled roughly by their handlers.
Due to their stimulated aggressive behavior, pigs taking the drug are also more likely to be injured during transport.13 In a review of the evidence, the researchers found additional adverse effects depending on the dose:14
"The evidence presented in the current review demonstrates that RAC [ractopamine] fed pigs may be more difficult to handle at doses above 5 mg/kg and physiological responses and rates of non-ambulatory pigs may increase when RAC fed pigs are subjected to aggressive handling, especially at the 20 mg/kg dose."
Health Risks of Ractopamine to Humans
The Codex Alimentarius Commission approved a maximum residue level of ractopamine of 10 parts per billion (ppb) in pork and beef, 40 ppb in liver and 90 ppb in kidneys.15 Only one human study was reviewed in the setting of this international standard — a study that involved six men, one of whom dropped out of the study due to adverse effects.16 Further, according to CFS:17
"Data from the European Food Safety Authority indicates that ractopamine causes elevated heart rates and heart-pounding sensations in humans. Other examples of health problems include information from the Sichuan Pork Trade Chamber of Commerce in China, which estimates that between 1998 and 2010, 1,700 people were poisoned from eating pork containing ractopamine."
Further, two drugs similar to ractopamine — zilpaterol and clenbuterol — are banned by the Olypmics because they affect adrenalin. In 2010, a cycler in the Tour de France failed an antidoping test for clenbuterol, which he blamed on residues from eating meat.18
A study in the journal Talanta further explained, "The use of highly active beta-agonists as growth promoters is not appropriate because of the potential hazard for human and animal health."19 There's even data showing human intoxication after consuming liver or meat from cattle treated with beta-agonists.20
Warning labels on the drug also state the risks outright, the Cornucopia Institute reported, including telling handlers to wear gloves and protective gear when handling it:21
"'WARNING: The active ingredient in Topmax, ractopamine hydrochloride, is a beta-adrenergic agonist. Individuals with cardiovascular disease should exercise special caution to avoid exposure,' says the label for the turkey feed. "Not for use in humans. Keep out of the reach of children.
The Topmax 9 formulation (Type A Medicated Article) poses a low dust potential under usual conditions of handling and mixing. When mixing and handling Topmax, use protective clothing, impervious gloves, protective eye wear, and a NIOSH-approved dust mask.
Operators should wash thoroughly with soap and water after handling. If accidental eye contact occurs, immediately rinse eyes thoroughly with water. If irritation persists, seek medical attention."
Ractopamine Is Administered Right Before Slaughter
Part of what makes ractopamine so controversial is the timing of its use. While some other livestock drugs are stopped in the weeks leading up to slaughter, giving some time for them to dissipate from the animal's system, ractopamine is administered in the weeks before slaughter.
When the U.S. Agricultural Research Service tested more than 1,000 pork kidney samples for four veterinary drugs, including ractopamine, the drug was detected in 22 percent.22 A Consumer Reports investigation also found detectable levels of ractopamine in about one-fifth of 240 pork products tested. The drug was also said to negatively affect the taste and tenderness of the meat.23
"While levels we found were below U.S. and international limits, Consumers Union, the policy and action arm of Consumer Reports, calls for a ban on the drug, citing insufficient evidence that it is safe," Consumer Reports noted.24 There is also evidence that the drug is present in manure, which is applied as fertilizer to soil, making it likely that it is also entering waterways, with unknown effects.25
However, research suggests the drugs may have endocrine-disrupting effects on marine species, including Japanese rice fish, or medaka.26 At least one study also showed ractopamine may be taken up by alfalfa and wheat from contaminated soil, at varying quantities depending on the concentration in soil.27
'Natural' Meat May Come From Ractopamine-Treated Animals
In a survey of more than 1,700 adults who said they try to eat healthy at least some of the time, 45% said they are likely to look for options that are all natural.28 When it comes to meat, however, the U.S. Department of Agriculture (USDA) allows the claim to be made for products that contain no artificial ingredients and minimal processing.
This means the meat may contain drugs, including antibiotics, hormones and preservatives, and still legally claim it's natural on the label, misleading consumers. The Animal League Defense Fund (ALDF) filed a lawsuit against meat giant Hormel for this very reason, alleging that the company engaged in potentially misleading advertising by using a Natural Choice label.
It turns out that the pigs used for Hormel Natural Choice products are the same as those used to create their spam, lunchmeat and bacon products, most of which consumers would not regard as natural. Further, most of the pigs have no access to the outdoors, and some products labeled Natural Choice came from pigs that received antibiotics and ractopamine.
The Superior Court of the District of Columbia dismissed ALDF's lawsuit, however, because the labeling is legal under the USDA definition of natural. "There's a difference between what's legal and what's ethical," Nikolas Contis, a senior partner at brand consultant PS212, told Farm Progress. "I think it's unethical. They know the words are misleading."29
ALDF attorney David Muraskin added, "It's a massive attempt to manipulate and dupe the consumer to purchase something they have no intention to purchase."30
Where to Find Truly Natural Pork
At one time, all pigs raised on U.S. family farms were heritage pigs, accustomed to roaming on pasture and in forests. The pigs don't take well to confinement conditions, however, and were soon replaced by commercial pigs bred to grow fast and tolerate crowding.
Whereas commercial pigs reach market weight in about six months, heritage pigs take about a year to do so. They're raised by a number of small farms, which typically sell the meat through farmers markets, food co-ops and occasionally to restaurants or niche markets.
The USDA doesn't define heritage breeds of pork, but the Livestock Conservancy defines them as heritage breeds if they have a long history in the U.S., are of noncommercial stock, thrive outdoors and on pasture and are purebred animals of their breed, according to Civil Eats.31
If you choose to eat pork, I encourage you to avoid CAFO meats and instead either buy your meat direct from a trusted grass fed farm raising heritage breeds or look for the American Grassfed Association (AGA) logo, a much-needed grass fed standards and certification for American-grown grass fed meat and dairy.32
The AGA standard allows for greater transparency and conformity33 and is intended to ensure the humane treatment of animals and meet consumer expectations about grass fed meat and dairy, while being feasible for small farmers to achieve.
The AGA pastured pork standards include a forage-based diet derived from pasture, animal health and welfare, no antibiotics and no added growth hormones. Because of the atrocious state of theCAFO pig industry, grass fed heritage pork is the only pork you should eat. At the very least, if you can't find grass fed heritage pork, choose organic pork products, as ractopamine is not allowed in organic foods.
Monday, April 29, 2019
Sunday, April 28, 2019
Friday, April 26, 2019
Wednesday, April 24, 2019
Tuesday, April 23, 2019
Why Treat Cancer with Bicarbonate? Baking Soda Rescues Circadian Oscillation
Published on April 19, 2019
In terms of importance, appropriate bicarbonate levels are just as crucial as hydration, proper breathing (getting enough oxygen) staying warm, maintaining healthy pH (high oxygen status) and optimal nutrition. What we learn here changes the face of modern oncology because it is now clear, after the most recent research presented below, that bicarbonates should be included in all cancer patients protocols.
Ten years ago I wrote Sodium Bicarbonate – Rich Man’s Poor Man’s Treatment For Cancer. Since then, many researchers have sustained the assertion that it would be deplorable to leave this most basic substance out of anyone’s cancer treatment, even if one is using toxic chemotherapy and radiation.
I am not suggesting that anyone should follow Dr. Tullio Simoncini’s bicarbonate treatments administered intravenously. There is little flexibility in the blood for pH changes. Simoncini got into serious trouble for over alkalizing the blood of a patient, a condition that easily ends in death. Using oral and transdermal administration is preferable not only because of safety issues but also because one can get bicarbonate to all the cells in the body, which is impossible if one adds bicarbonate directly into the blood.
Simple baking soda is the ideal medicine for mitigating radiation exposure. It helps clear the kidneys of uranium toxicity. It certainly has been used when chemotherapy is administered and during cardiac arrest. It is also the treatment of choice for fungus infections. It addresses CO2 deficiencies. It helps in efforts to detoxify the body of the thousands of chemicals infecting us. It is an excellent antiviral pulling the rug out from under the little critters that are sensitive to pH changes. Most viruses are pH sensitive.
Breakthrough Bicarbonate Cancer Research
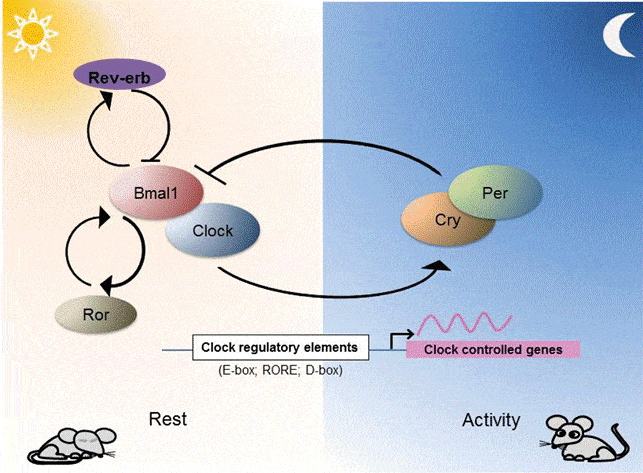
Now we have more reason than ever to focus on bicarbonate therapy for cancer. A new Ludwig Cancer Research study said, “If you want to clean cancer’s clock—that is, defeat it decisively—you may want to really clean it—that is, restore it to proper working order. Only cancer cells that remain active, in sympathy with the tick-tock of their internal circadian mechanisms, remain susceptible to cancer therapies. So, how might these circadian mechanisms be kept in motion? Sodium bicarbonate now promises to awaken cancer cells that have gone to sleep deep insidetumors, where oxygen deprivation and acidic conditions go hand in hand. By buffering against acidification, sodium bicarbonate rescues circadian oscillation.”
Learn a system of medicine that is safe, simple and affordable! Discover Dr Sircus Natural Allopathic Medicine »
You can think of the circadian cycle as a sort of internal clock of your body that regulates many different mechanisms. The cycle starts over about every 24 hours (hence the term “circadian”, coming from Latin circa “about” and dies “day”), and among its many functions, it tells cells when they’re supposed to produce and consume nutrients. Healthy cells are normally allowed to do so for about 12 hours a day, in order to prevent them from being overwhelmed by a flood of excessive nutrients.”
In order to keep dividing indefinitely, cancer cells cannot afford the periodic fasting imposed by the circadian rhythm; before they can divide, they need to grow, and in order to grow, they need nutrients. Therefore, furiously paced division requires furiously paced nutrient intake. Hence, all cancers evolve strategies to get around the circadian limitation.”
Acidification, the Ludwig researchers report, essentially disengages some of the elements that keep the cell’s circadian clock ticking. This acid-mediated effect, the researchers assert, can be reversed by sodium bicarbonate. When they gave tumor-xenografted mice water that had been supplemented with sodium bicarbonate, the acidity of hypoxic patches in the tumors was neutralized.
The study, presented May 31 2018, in the journal Cell, in an article entitled ‘Acid Suspends the Circadian Clock in Hypoxia through Inhibition of mTOR’. “Unexpectedly, we uncover a profound disruption of the circadian clock and diurnal transcriptome when hypoxic cells are permitted to acidify to recapitulate the tumor micro-environment,” the article states. “Buffering against acidification or inhibiting lactic acid production fully rescues circadian oscillation.”
The study’s authors, led by Chi Van Dang, M.D., Ph.D., scientific director of the Ludwig Institute for Cancer Research, conclude that acid produced during the cellular metabolic response to hypoxia suppresses the circadian clock through diminished translation of clock constituents. Dr. Dang, who is also a professor in the Molecular and Cellular Oncogenesis Program at The Wistar Institute said, “But if you add baking soda to the drinking water given to those mice, the entire tumor lights up with mTOR activity. The prediction would be that by reawakening these cells, you could make the tumor far more sensitive to therapy.”
“We started out with a question about oxygen starvation and the circadian clock, and we ended up discovering a new mechanism by which acidic conditions in tissues shut off a lot of things—including the cell’s molecular clock,” muses Dang.
The finding that something as simple as baking soda could reverse this effect and render quiescent cancer cells susceptible to cancer therapies excites Dang. “The concept is so easy,” he says. “It’s not some $100,000 per year drug. It’s literally just baking soda.” Using MRI, medical scientists have seen how much of tagged bicarbonate is converted into carbon dioxide within tumors. In more acidic tumors, more bicarbonate is converted into carbon dioxide.
Cancer and pH
Virtually every cancer patient will benefit from the use of sodium bicarbonate because it treats the low oxygen acid conditions universally found in cancer patients. Sodium bicarbonate shrinks tumors. (Bicarbonate inhibits spontaneous metastases (Robey 2009). ‘Bicarbonate Increases Tumor pH and Inhibits Spontaneous Metastases’ according to medical scientists. NaHCO3 therapy significantly reduced the formation of hepatic metastases following intrasplenic injection, suggesting that it did inhibit extravasation and colonization.
“Studies conducted at the University of Bari in Italy clearly demonstrated that a hallmark of all tumors, regardless of their origin or background, is their acidic environment. In fact, tumor progression increased with an acidic pH and hypoxia, or a low oxygen level,” writes Dr. Veronique Desaulniers. The perturbation in pH dynamics rises very early in carcinogenesis and is one of the most common patho-physiological hallmarks of tumors.”
“The results of a study suggest that tumor cells do, indeed, perform niche engineering by creating an acidic environment that is non-toxic to the malignant cells but, through its negative effects on normal cells and tissue, promotes local invasion.”
Tumor invasion did not occur into regions with normal or near-normal pH. Immuno-histochemical analyses revealed that cells in the invasive edges expressed the glucose transporter GLUT-1 and the sodium-hydrogen exchanger NHE-1, both of which were associated with peritumoral acidosis. In support of the functional importance of our findings, oral administration of sodium bicarbonate was sufficient to increase peritumoral pH and inhibit tumor growth and local invasion in a preclinical model, supporting the acid-mediated invasion hypothesis.
Increased systemic concentrations of pH buffers leads to reduced intratumoral and peritumoral acidosis. Oral NaHCO3 selectively increased the pH of tumors and reduced the formation of spontaneous metastases in mouse models of metastatic breast cancer. NaHCO3 therapy also reduced the rate of lymph node involvement and significantly reduced the formation of hepatic metastases. Acid pH was shown to increase the release of active cathepsin B, an important matrix remodeling protease.
Cancer, Infections and Baking Soda
Oncologist Dr. Tullio Simoncini was persecuted for his use of sodium bicarbonate for cancer patients because he laid attention to the relationship between cancer and fungus. It needs to be remembered when connecting cancer with fungus that Cancer—always believed to be caused by genetic cell mutations—can in reality be caused by infections from viruses, bacteria, and fungi.
“I believe that, conservatively, 15 to 20% of all cancer is caused by infections; however, the number could be larger—maybe double,” said Dr. Andrew Dannenberg, director of the Cancer Center at New York-Presbyterian Hospital/Weill Cornell Medical Center. Dr. Dannenberg made the remarks in a speech in December 2007 at the annual international conference of the American Association for Cancer Research so it is another reason to treat cancer with a bicarbonate, which has strong anti-infection effects.
Conclusion
Greater knowledge of the mechanisms that contribute to the survival of tumor cells is key to vanquishing them. Shutting down the circadian oscillation is one principle strategy that cancer cells employ to thrive and sodium bicarbonate fully rescues circadian oscillation. Thus it is time for mainstream oncologists to wake up to what medical science has to say about bicarbonates. There is just too much benefit and practically zero downside to bicarbonates. Bicarbonate has long been used in ICU, emergency departments and in ambulances. There is no reason it should not be employed for all cancer patients.
Below is the product that I use daily to get my sodium and potassium bicarbonate. In the next chapter, which merges the use of bicarbonates with fasting, we will introduce magnesium bicarbonate, which one can add to all one’s water. Magnesium bicarbonate is the strongest form of bicarbonate, has no salt though it does not compare in terms of cost with sodium or potassium bicarbonate.

pH ADJUST may be used to increase salivary and urinary pH; counteract overly acidic conditions in the digestive tract, blood, and kidneys; and to supplement the body with the minerals potassium, magnesium, and sodium. The product contains (in powder form) potassium bicarbonate, magnesium carbonate, potassium glycinate, and sodium bicarbonate. Each serving (about ¼ tsp) contains about 300 mg of bicarbonate, 260 mg of carbonate, 142 mg of potassium, 105 mg of magnesium, 48 mg of sodium, and 100 mg of glycine.
Potassium is excellent for heart health and in potassium sufficient people, it reduces risk of strokes. Potassium also helps maintain healthy digestive and muscular function.
You Are a Guinea Pig — Undisclosed GMO Soybean Oil Released
- April 23, 2019

STORY AT-A-GLANCE
- A gene-edited soybean oil created by biotech company Calyxt was picked up by its first user — a Midwest company with both restaurant and foodservice locations, which is using it for frying as well as in dressings and sauces
- Calyxt’s soybean oil, Calyno, contains two inactivated genes, resulting in an oil with no trans fats, increased heart-healthy oleic acid and a longer shelf life
- Along with refusing to identify the buyer of its gene-edited high-oleic soybean oil, Calyxt is marketing its product as “non-GMO”
- Although they’re genetically engineered, gene-edited foods are not marketed as GMOs, nor are they labeled as such
- Because they contain no foreign genetic material, foods produced via gene-editing are not subject to regulation by the U.S. Department of Agriculture — although an advisory board recommended gene-edited foods could not be labeled organic
The first commercially available gene-edited food is now on the market, but consumers won't know where it's being sold or if they've eaten food that contains it. The product, a gene-edited soybean oil created by biotech company Calyxt, was picked up by its first user — a Midwest company with both restaurant and foodservice locations, which is using it for frying as well as in dressings and sauces.1
Calyxt's soybean oil, Calyno, contains two inactivated genes, resulting in an oil with no trans fats, increased heart-healthy oleic acid and a longer shelf life. Along with refusing to identify the buyer of its gene-edited high-oleic soybean oil, Calyxt is marketing its product as "non-GMO," although it's clearly genetically engineered.
Using Semantics to Hide GMOs
Although they're genetically engineered, gene-edited foods are not marketed as GMOs, nor are they labeled as such. The difference comes down to a matter of semantics.
Calyxt used Transcription Activator-Like Effector Nuclease (TALEN) to find and edit DNA sequences in the making of its soybean oil, a process a company spokesperson went so far as to say could happen in nature. Speaking to Forbes, they stated:2
"[U]nlike GMOs, we simply edit existing genes within crops using our technology to speed up a process that otherwise could have happened in nature.
Through this process, we're able to provide outcomes quickly, efficiently and cost-effectively for the American people so that they can have healthier food ingredients without compromising the taste of what they already love. No foreign DNA is added to the product."
Unfortunately, they've also succeeded in introducing a genetically engineered oil to the U.S. food supply without the American people's knowledge or consent. The appeal to food manufacturers is clear: Calyxt's oil, with its zero trans fats and long shelf life, will appeal to companies eager to replace trans fats.
In an announcement through Bloomberg,3 Calyxt said this high-oleic oil contains "approximately 80 percent oleic acid and up to 20 percent less saturated fats." Calyxt also said they'd just completed the company's first commercial sale of a "premium," high oleic soybean meal as a livestock food additive that would be an "added benefit" for the livestock.
But the public may have another take on the matter, especially as many increasingly seek out real, whole foods in lieu of GMOs. One survey found only 32 percent of Americans are comfortable with GMOs in their food.4
By hiding behind the label of gene editing, they can pass off their genetically engineered Calyno oil as natural when it's clearly not. More than 100 farmers in the Midwest are reportedly growing Calyxt's high-oleic soybeans on more than 34,000 acres.5
Gene-Edited Chickens Created
If eating gene-edited soybean oil has you feeling like a guinea pig, you may be equally uneasy to know that gene-edited chickens are also a thing. At the Roslin Institute at the University of Edinburgh in Scotland, chickens have been modified to resist flu, which spreads rapidly among CAFO (concentrated animal feeding operation) birds and has the potential to be transmitted to humans.
In order to create the transgenic chickens, scientists used the gene-editing technology known asCRISPR, or Clustered Regularly Interspaced Short Palindromic Repeat. They targeted part of the ANP32 gene, which codes for a protein that flu viruses depend on,6 and cells without the gene were impervious to the flu.
The Roslin Institute has also used gene editing to create pigs that are resistant to a disease called Porcine Reproductive and Respiratory Syndrome, or PRRS, edits that are permanent and passed down to other generations. Other companies are using gene editing to remove genes that grow horns in dairy cattle, therefore allowing them to bypass the inhumane process of removing horns fromCAFO cattle with no pain relief.
There is talk that first using gene editing to ease animal suffering or fight agricultural disease could soften regulators' stance and create a more favorable profile to the public.7 But ultimately the technology will inevitably be used increasingly for the purpose of profits.
Case in point, one study funded by the U.S. Department of Agriculture has added the SRY gene to cattle, which results in female cows that turn into males, complete with larger muscles, a penis and testicles, but no ability to make sperm.8 Male (or male-like) cattle are more valuable to the beef industry because they get bigger, faster, allowing companies to make greater profits in less time.
The U.S. Food and Drug Administration (FDA) proposed classifying animals with edited or engineered DNA as drugs, prompting backlash from the biotech industry,9 which doesn't want such foods labeled. The Roslin Institute has also launched a survey to gauge people's views on gene-editing and whether or not they would eat gene-edited animals.10
However, because they contain no foreign genetic material, foods produced via gene-editing are not subject to regulation by the U.S. Department of Agriculture (USDA) — although an advisory board recommended gene-edited foods could not be labeled organic — or other regulatory agencies.
Gene Editing Used to Create High-Fiber Wheat, Altered Potatoes
To date, gene editing has been used not only to produce soybeans with altered fatty acid profiles, but also potatoes that take longer to turn brown and potatoes that remain fresher longer and do not produce carcinogens when fried. Other uses for gene-editing in foods include the creation of low-gluten wheat, mushrooms that don't turn brown and tomatoes that can be produced in areas with shorter growing seasons.
The technology may even be used to create plants that withstand droughts and diseases or seeds that can be customized to unique growing conditions. Calyxt has also developed a high-fiber wheat that has been declared a "nonregulated article," by the USDA's Biotechnology Regulatory Services of the Animal and Plant Health Inspection Service (APHIS). The wheat is in Phase 1 of development and may launch as early as 2020 or 2021.11
Calyxt has lofty plans for expansion and is also working on gene-edited cold-storable potatoes andcanola oil with improved composition, and hopes to eventually create products with many gene-edited targets in one. According to the company:12
"In particular, over time we may explore opportunities to apply our commercial strategy elsewhere around the world and leverage our North American products and footprint to target geographies where there are unmet consumer or farmer needs.
We also intend to explore the ability to add value through our existing product candidates once they are commercialized by combining traits in the same crop, which may allow us to create products with additional benefits without adding significant cost."
Gene Editing Isn't Always Precise, Carries Significant Risks
While gene-edited foods have already been released into the food supply, there safety is largely unknown. What is known, however, is that gene editing isn't a perfect science, and off-target edits could cause unintended changes to plant DNA, with consequences that could include growth disturbances, exposure to plant diseases or the introduction of allergens or toxins.13
In animals, gene editing has led to unexpected side effects, including enlarged tongues and extra vertebrae.14,15 Often researchers don't know the extent of a gene's functions until they attempt to tweak it, and something like an extra vertebra reveals itself. Speaking with Yale Insights, Dr. Greg Licholai, a biotech entrepreneur, explained some of the very real risks of CRISPR and other gene-editing technologies:16
"One of the biggest risks of CRISPR is what's called gene drive, or genetic drive. What that means is that because you're actually manipulating genes and those genes get incorporated into the genome, into the encyclopedia, basically, that sits within cells, potentially those genes can then be transferred on to other organisms.
And once they're transferred on to other organisms, once they become part of the cycle, then those genes are in the environment.
That's probably the biggest fear of CRISPR. Humans manipulating the genetic code, and those manipulations get passed on generation to generation to generation. We think we know what we're doing, we think we're measuring exactly what changes we're doing to the genes, but there's always the possibility that either we miss something or our technology can't pick up on other changes that have been made that haven't been directed by us.
And the fear then is that those changes lead to antibiotic resistance or other mutations that go out into the population and would be very difficult to control. Basically creating incurable diseases or other potential mutations that we wouldn't really have control over."
Japan Follows US, Says Gene Edited Foods Are Safe
While the European Union has ruled that gene edited crops must go through the same approval process as GMOs, Japan recently concluded that such foods could enter the consumer marketplace without any safety studies. "There is little difference between traditional breeding methods and gene editing in terms of safety," Hirohito Sone, an endocrinologist at Niigata University, stated.17
However, in an interview with GM Watch, Michael Antoniou, a London-based molecular geneticist, explained that significant changes could occur due to genetic editing, in both agricultural and medical contexts, necessitating long-term safety and toxicity studies. He explained:18
"Many of the genome editing-induced off-target mutations, as well as those induced by the tissue culture, will no doubt be benign in terms of effects on gene function. However, many will not be benign and their effects can carry through to the final marketed product, whether it be plant or animal …
Thus not only is it necessary to conduct whole genome sequencing to identify all off-target mutations from CRISPR-based genome editing, but it is also essential to ascertain the effects of these unintended changes on global patterns of gene function.
… In addition, it is important to acknowledge that the targeted intended change in a given gene may also have unintended effects. For example, the total disruption or modification of an enzyme function can lead to unexpected or unpredictable biochemical side-reactions that can markedly alter the composition of an organism, such as a food crop.
The compositional alterations in food products produced with genome editing techniques will not be fully revealed by the molecular profiling methods due to the current inherent limitations of these techniques. So it is still necessary to conduct long-term toxicity studies in established animal model systems. In the absence of these analyses, to claim that genome editing is precise and predictable is based on faith rather than science."
Organic Foods Are Not Gene Edited
Without a label requirement, there's no way for consumers to know whether they're eating gene-edited soybean oil — or one of the many future gene-edited products likely to hit the market. For now, however, gene-edited foods cannot be labeled organic, which is one more reason why seeking out organic and, even better, biodynamic foods, is so important.
This may not be the case forever, though, as some organic proponents have claimed gene editing falls within the realm of organic if used in a way that mimics nature.19 That being said, when the National Organic Standards Board voted (unanimously) to add CRISPR to the excluded methods list, one board member said:20
"It very clearly is a GMO [method] and has no field history of improved varieties to evaluate what unintended effects the technology might have on the environment, just like all of the GMOs released into the environment have had unintended effects that don't show up for a number of years."
Whether the U.S. government will ultimately decide to classify gene-edited foods similarly to GMOs or conventional foods, or to allow them under the organic label, remains to be seen, but for now the best way to avoid gene-edited foods, if you so choose, is to purchase organic.
Subscribe to:
Posts (Atom)
